PLEASE SELECT THE DESTINATION COUNTRY AND LANGUAGE :
HandyRay(LMT-3000)

Brand new device for the comfortable and safe blood collection. HandyRay makes the capillary blood collection comfortable and safe with low pain, no risk of cross infection and fast regeneration of skin.




Product specification
| Medical device class | Wavelength | Laser level range | Repetition | Battery chager |
|---|---|---|---|---|
| KFDA Ⅲ | 2940nm | 1~5 Step | Single shot operationd | 3.7V |
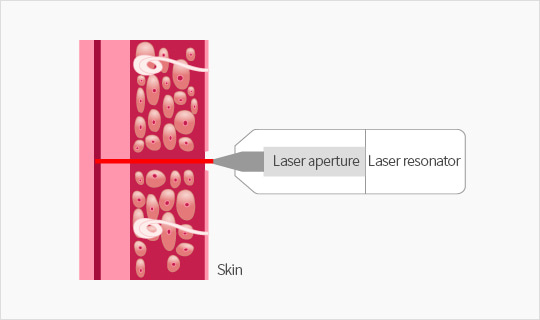
Operation principle
- - Using the safe laser( Er: YAG laser) in compliance with laser and medical device safety standard
- - Er: YAG laser generates the high energy when the laser contacts the skin
- - High energy generated by the laser ablates the skin and makes a small hole in a very short time (less than1/10000 second) and the capillary blood can be collected through the hole
Benefits
- - User-friendly: Low pain and easy to use for blood sampling time
- - Safe: HandyRay uses the safe laser source,
which fully complies with the international safety standard for the laser - - Hygienic: Disinfecting the skin due to high energy laser
How to use
01 Press and hold the power button for 3~4s

02 Choose a proper laser level

03 Put single-use cap on to protect the device

04 Massage the tip of your finger

05 Put the device on spot where blood sample

06 Collect the blood

Cautions for use


Must use the single-use cap to protect the device.
As the reuse of the single-use cap might result in failure of the blood collection, a user should not reuse the single-use cap.


Do not move or put your finger away from the laser light while using the laser.
Firmly press the device onto your finger to make sure that the laser aperture contacts the finger tightly.
Keep contact between your finger and the device, use the laser and remove the device after 1~2 seconds.

Wipe the lens in case the device is contaminated.
It is recommended to clean the lens of the device on a regular basis.


In case of malfunction, push the reset button to initialize the device before use.
Product constitution
Laser lancing device

User’s Manual

Charger

Pouch

Disposable Cap Package(100pcs)

Quick Guide

Product Certification
01Company Certification

2017. 06
KGMP

2012. 02
Europe ISO 13485 certificate

2016. 04
Venture company certificate

2012. 04
Company R&D center registration
02Product Certification

2017. 07
KFDA approval
(Medical device class 3)
2018. 03
FDA (510k) approval

2016. 02
CE Marking

2019. 07
NET certificate
This product is a medical device. Please make sure to read precaution and manual before its use."
Certified advertisement review No. : 2017-110-35-2348

